Bayesian in comprehensive meta analysis
Linaclotide, a peptide homolog of Escherichia coli ST toxin, interacts with the guanylate cyclase C (GC-C) located in the enterocyte apical membrane. However, up to 50% of patients report that they are not completely satisfied with current treatment due to inefficient relief from constipation, the side effects of drugs (e.g., bloating and abdominal pain), the lack of predictability of laxative action, and partial improvement of the quality of life. There are many choices available for the treatment of CC, including changes in defecation habits, increase in fiber intake, and several drugs, such as laxatives and systemically active agents. Thus far, the risk factors for CC identified are advanced age, female, low socioeconomic status, low parental education rates, decreased physical activity, certain medications, stressful life events, physical and sexual abuse, and depression. There has been a steady and significant increase in the proportion of ambulatory care related to this disorder, and it poses a heavy economic burden for healthcare systems. Approximately one in five people presented with CC symptoms will seek medical help. The estimated incidence of CC ranges from 11% to 20%. In contrast, poor quality of life can aggravate CC symptoms. The most widely used diagnostic criteria of CC are the Rome criteria, and the latest is Rome IV, created in 2016, which distinguishes functional constipation from irritable bowel syndrome with constipation (IBS-C).ĬC negatively affects the quality of life, comparable with chronic diseases, such as chronic obstructive pulmonary disease, diabetes, and depression. CC is characterized by various bowel symptoms, including infrequent bowel movement, hard stools, excessive straining to defecate, a sense of anorectal blockage, anal digitation, and a sense of incomplete evacuation. IntroductionĬhronic constipation (CC) may be primary (idiopathic or functional) or secondary to several disorders or medications. High dose of linaclotide could be more effective and safer for CC patients, which need more trials to confirm in the future. Additionally, no more any adverse events occurred in the higher linaclotide dose group (RR: 0.91 95% CrI: 0.60–1.38) indirectly compared with other dose groups. However, no significant difference presented in the CSBM responder rate by pairwise comparisons of the different dose groups.
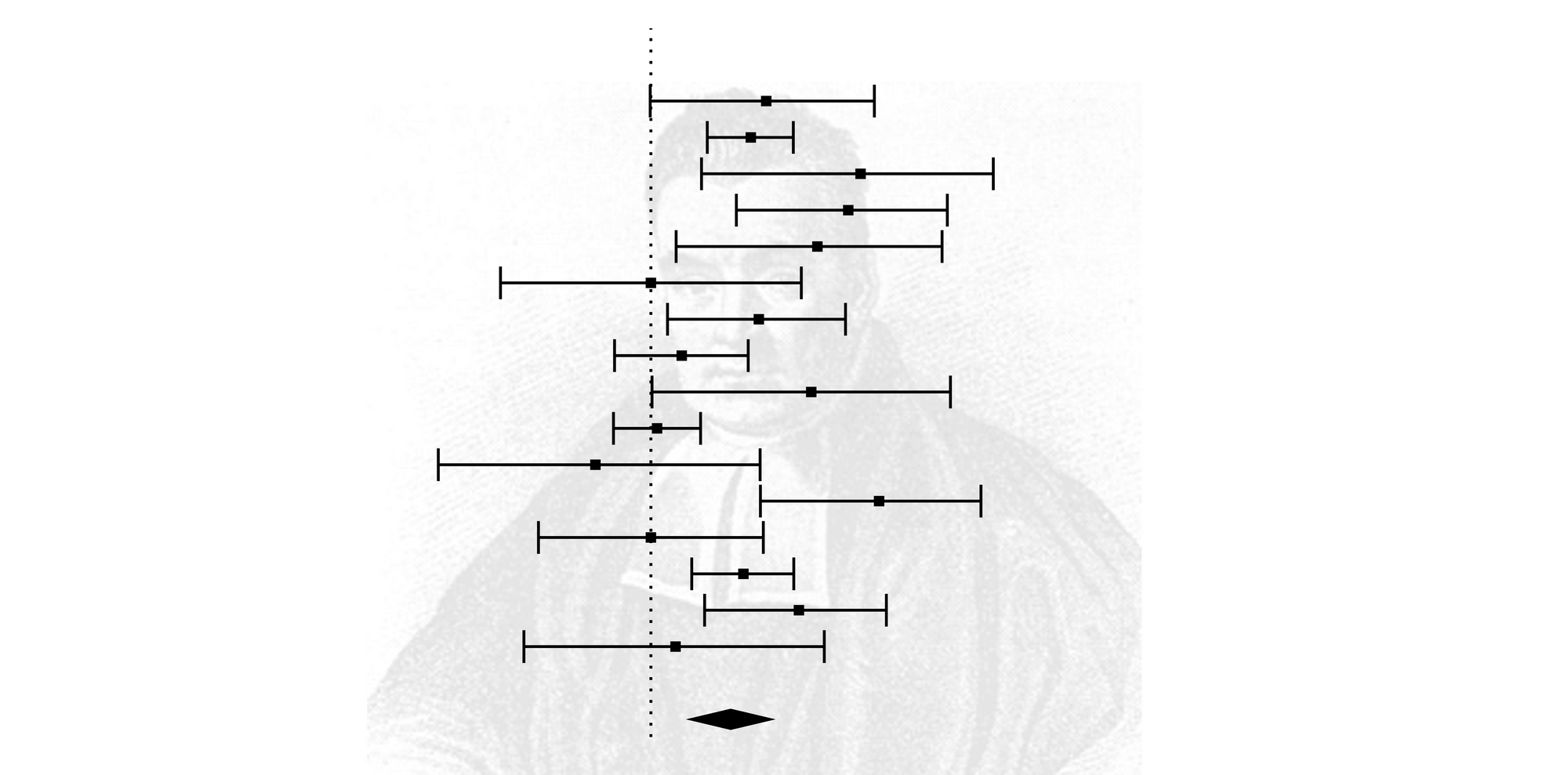

Bayesian analysis showed the high-dose group obtained the maximum CSBM responder rate (OR: 4.94 95% credible interval (CrI): 3.22–7.79 probability rank = 0.87) indirectly compared with extremely low-dose, low-dose, and medium-dose groups. A significantly enhanced number of completely spontaneous bowel movement (CSBM) responders were found in the extremely low-dose group (OR: 2.94 95% CI: 1.98–4.34 ), the low-dose group (OR: 3.24 95% CI: 2.44–4.31 ), the medium-dose group (OR: 3.08 95% CI: 1.46–6.50 ), and high-dose group (OR: 4.79 95% CI: 3.04–7.54 ).

Seven studies with 4,107 patients were eligible.
#BAYESIAN IN COMPREHENSIVE META ANALYSIS SOFTWARE#
A comprehensive search was conducted, and STATA16 software was used for data analysis. The meta-analysis aimed to assess the efficacy and safety of linaclotide doses ranging from 62.5 μg to 600 μg for CC patients. It is ambiguous whether a higher dose of linaclotide provides higher efficacy for chronic constipation (CC) patients.
